iPSCs
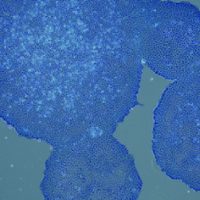
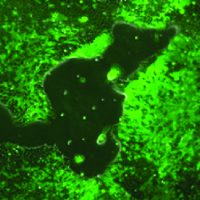
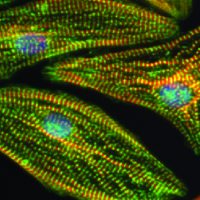
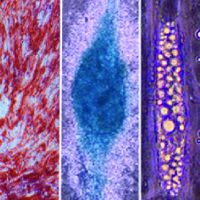
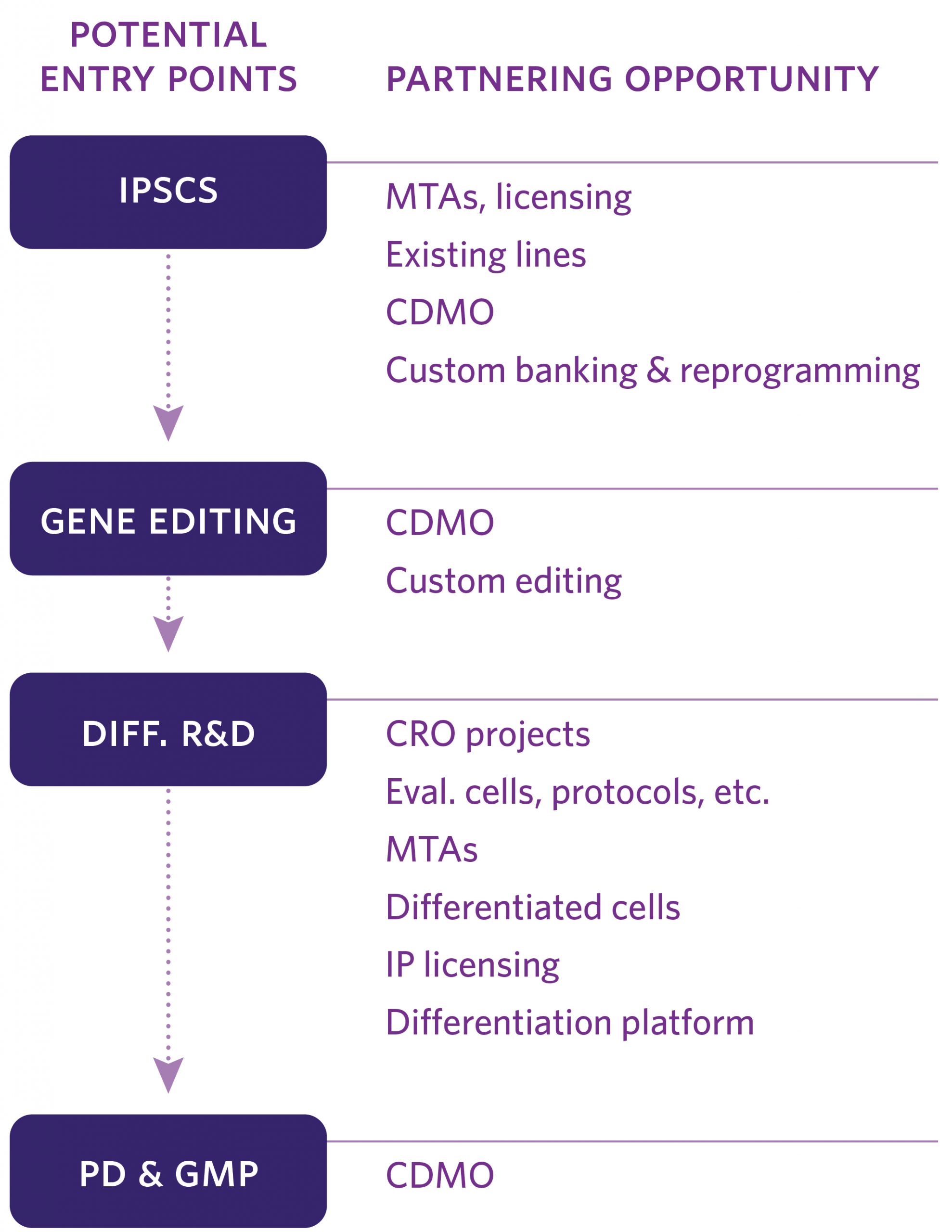
Human induced pluripotent stem cells (iPSCs) enable next-generation cell therapies for a variety of disease areas, most notably in cell replacement and anti-cancer therapy. Our end-to-end value chain offering links our in-house GMP iPSC lines with advanced manipulation platforms and GMP manufacturing (see adjacent figure for partnering opportunities).
Our GMP iPSC lines of EU and US origin are extensively characterized and available off-the-shelf for evaluation under R&D conditions as well as for GMP licensing. Moreover, Catalent scientists have developed GMP-friendly, state-of-the-art workflows for iPSC manipulation addressing critical bottlenecks in each case that are simple, robust, weekend-free, and produce highly pure differentiated cell populations. (click each cell line to navigate to its specific page):
The certified reprogramming and iPSC banking workflow is also offered as a universal platform for custom banking and QC testing based on in-house or external cell lines. Furthermore, beyond the above editing and differentiation methodologies which are offered for licensing and as technical platforms for superior GMP manufacturing. Talk to your Catalent representative to discuss partnering with us to establish additional workflows.