- Home »
- Specialty »
- Plasmid DNA » Plasmid Manufacturing

Plasmid Manufacturing
 Catalent offers customized GMP plasmid DNA services from milligram to gram scale for use as raw materials in your viral and non-viral applications. Our experienced team will leverage Catalent’s high yielding and robust plasmid UpTempo℠ analytical and process platforms to support your program as it progresses from development phase towards late-stage studies and commercialization. Our integrated manufacturing services for plasmid DNA, viral vectors, and cell therapies provide our partners with a reliable, robust supply chain. A partnership with Catalent for plasmid DNA and viral vector manufacturing facilitates a streamlined process with consistent quality, and allows you to leverage our expertise in developing scalable, commercial-ready processes and our EMA and FDA approved commercial viral vector facility. Catalent is committed to helping our partners get their advanced therapies to patients, faster.
Catalent offers customized GMP plasmid DNA services from milligram to gram scale for use as raw materials in your viral and non-viral applications. Our experienced team will leverage Catalent’s high yielding and robust plasmid UpTempo℠ analytical and process platforms to support your program as it progresses from development phase towards late-stage studies and commercialization. Our integrated manufacturing services for plasmid DNA, viral vectors, and cell therapies provide our partners with a reliable, robust supply chain. A partnership with Catalent for plasmid DNA and viral vector manufacturing facilitates a streamlined process with consistent quality, and allows you to leverage our expertise in developing scalable, commercial-ready processes and our EMA and FDA approved commercial viral vector facility. Catalent is committed to helping our partners get their advanced therapies to patients, faster.
UPTEMPO℠ PLASMID PLATFORM PROCESS
Catalent has developed a process and analytical platform to reduce development time and accelerate the transition of programs from a pre-IND phase to the clinic and bring these curative therapies to patients. The platform process has been designed to accommodate a wide range of different plasmid sequences and is characterized by its high yield, capable of producing as much as 0.5 g of purified plasmid DNA per liter of fermentation culture. The manufacturing process is fully disposable which further reduces the turn-around time and mitigates the risk of cross contamination. Catalent’s analytical platform and set of specifications are designed to ensure that the plasmid purity profiles are aligned with program requirements and comply with CGMP regulatory guidelines.
The UpTempo℠ plasmid DNA platform process integrates the creation of a cell bank, upstream and downstream activities, and analytical testing. The process starting point is a fully characterized E. coli cell bank that is well suited to produce the plasmid of interest. The E. coli cells containing the plasmid to be produced are cultured, and plasmid copy number is expanded through a high yielding fermentation fed-batch process. Using this fermentation strategy, Catalent reaches plasmid DNA yields that can exceed 1 g of plasmid DNA per liter of cell culture medium.
The plasmid-containing cells are lysed through a controlled alkaline treatment, which is essential for maximizing product recovery while preserving the integrity of the plasmid molecules. Catalent has engineered an in-line cell lysis unit operation that provides rigorous process control, effectively clears E. coli contaminants, and shortens the overall processing time.
The resulting lysate is then clarified by filtration, concentrated to a more manageable volume, and buffer exchanged by ultrafiltration/diafiltration (UF/DF) into a solution suitable for subsequent chromatography. A notable feature of the UpTempo℠ platform process is its reliance on a single chromatographic step to achieve the desired plasmid purity profile. This chromatographic unit operation clears any remaining E. coli residual contaminants as well as open circular and linear product-related isoforms. The eluted purified and high supercoiled content plasmid DNA is then formulated in the desired formulation buffer of choice and concentrated as required by UF/DF. The process concludes with a final 0.2 µm filtration step.
Alongside the circular plasmid DNA process supporting viral vector applications, Catalent has developed a process flow to produce linear plasmid DNA used as templates for the IVT production of mRNA molecules. Catalent has incorporated into its process, following chromatography, an enzymatic digestion unit operation to effectively linearize supercoiled enriched plasmid DNA.
Plasmids undergo thorough testing using Catalent’s set of analytical assays, which encompasses both compendial and qualified & validated assays. This analytical platform will confirm that the plasmids satisfy the necessary critical quality attributes as defined by regulatory guidelines and the specific needs of the program.
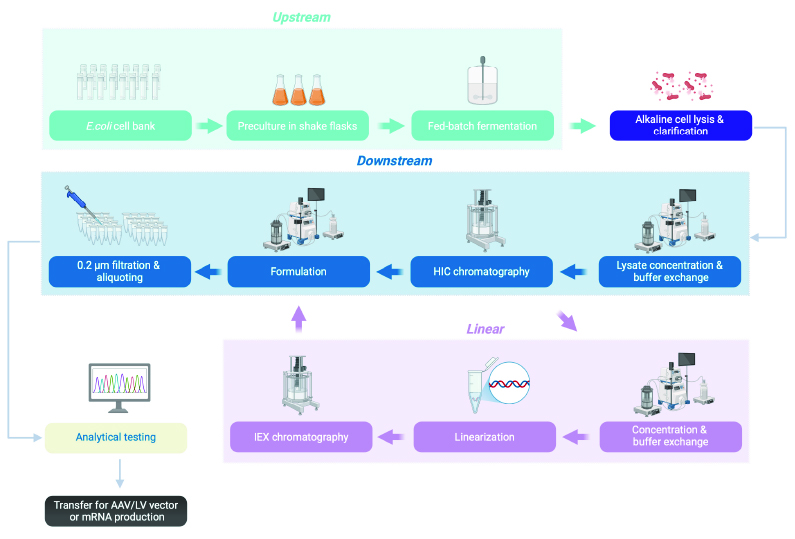
Figure 1: The plasmid DNA platform process workflow from E. coli cell bank fermentation through purification, analytical testing and transfer to the next step in advanced therapy production. Unit operations in purple refer only to purification of linear pDNA. Created in BioRender https://BioRender.com/y79e761
The plasmid DNA platform process workflow from E. coli cell bank fermentation through purification, analytical testing and transfer to the next step in advanced therapy production.
Upstream – Upstream processing begins with an E. coli cell bank that is first precultured in shake flasks and then subsequently grown by fed-batch fermentation. Upstream processing is followed by alkaline cell lysis and clarification.
Downstream – Downstream processing starts with lysate concentration and buffer exchange followed by purification by HIC chromatography and subsequent formulation, 0.2 µm filtration and aliquoting of the purified plasmid DNA.
Linear – When working with linear plasmid DNA, purified plasmid DNA obtained from HIC chromatography is subjected to concentration and buffer exchange, followed by linearization and subsequent purification by IEX chromatography.
The final aliquoted plasmid DNA is subjected to analytical testing followed by transfer for AAV/LV vector or mRNA production.
